
More than two million people worldwide are diagnosed with breast cancer each year. We look forward to realizing the full potential of this TROP2-directed antibody drug conjugate across breast cancer subtypes through our ongoing Phase III program, including two trials in patients with triple-negative breast cancer.” Ken Takeshita, MD, Global Head, Oncology R&D, Daiichi Sankyo, said: “The positive topline results from TROPION-Breast01 demonstrate the potential for datopotamab deruxtecan to become an important treatment option for patients with HR-positive, HER2-low or HER2-negative breast cancer in the second-line metastatic setting. 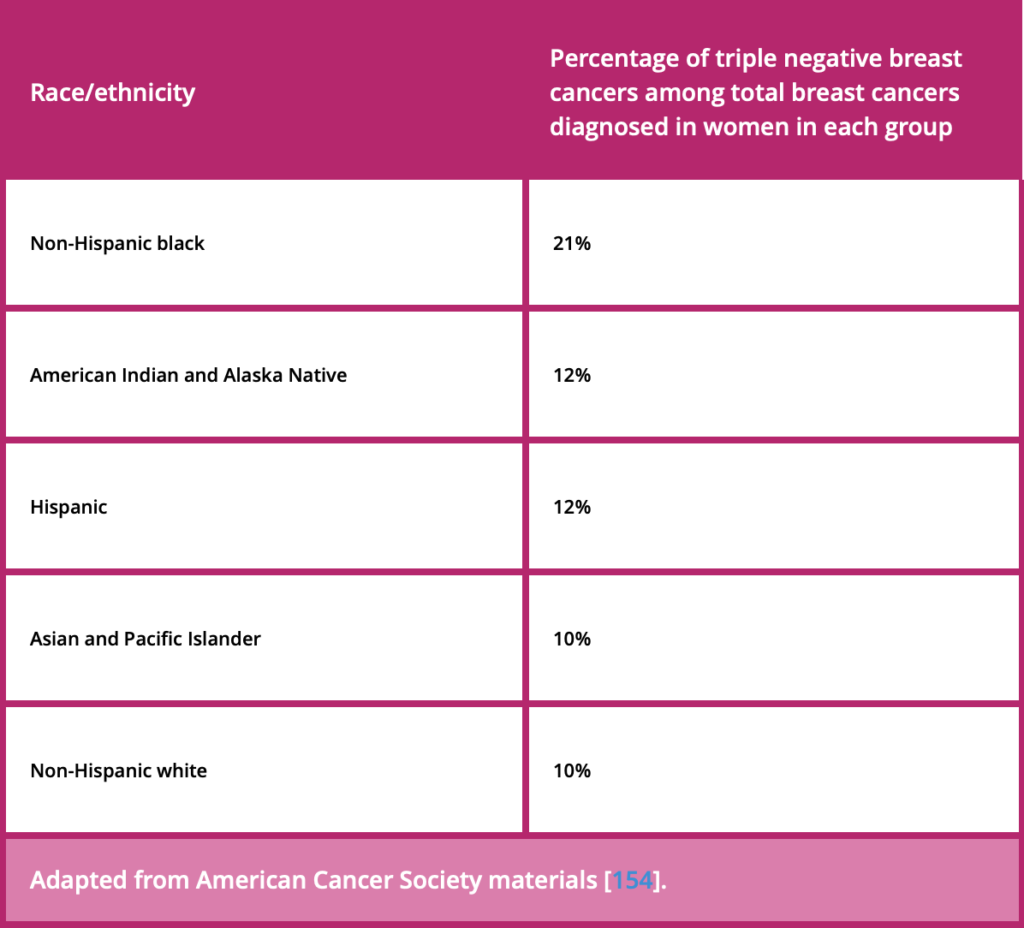
We are encouraged by these positive results.” Susan Galbraith, Executive Vice President, Oncology R&D, AstraZeneca, said: “Today’s TROPION-Breast01 news is a significant development for patients with HR-positive, HER2-low or negative metastatic breast cancer whose tumors have become insensitive to endocrine therapy and who currently face poor outcomes. All grade interstitial lung disease rates were low.ĭatopotamab deruxtecan is a specifically engineered TROP2-directed DXd antibody drug conjugate (ADC) being jointly developed by AstraZeneca and Daiichi Sankyo. The safety profile of datopotamab deruxtecan was consistent with previous clinical trials in breast cancer with no new safety signals identified. Data for OS were not mature at this interim analysis and the trial will continue as planned to assess OS. Positive high-level results from the TROPION-Breast01 Phase III trial showed datopotamab deruxtecan (Dato-DXd) demonstrated a statistically significant and clinically meaningful improvement for the primary endpoint of progression-free survival (PFS) compared to investigator’s choice of chemotherapy in patients with inoperable or metastatic hormone receptor (HR)-positive, HER2-low or negative (IHC 0, IHC 1+ or IHC 2+/ISH-) breast cancer previously treated with endocrine-based therapy and at least one systemic therapy.Ī trend in improvement for the dual primary endpoint of overall survival (OS) was observed for datopotamab deruxtecan versus chemotherapy. Plans for global regulatory submissions underway 
First Phase III results in breast cancer for AstraZeneca and Daiichi Sankyo’s datopotamab deruxtecan
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
